Toolbox Strategic Focus Area 1
Cataloguing and publishing descriptions of data sources
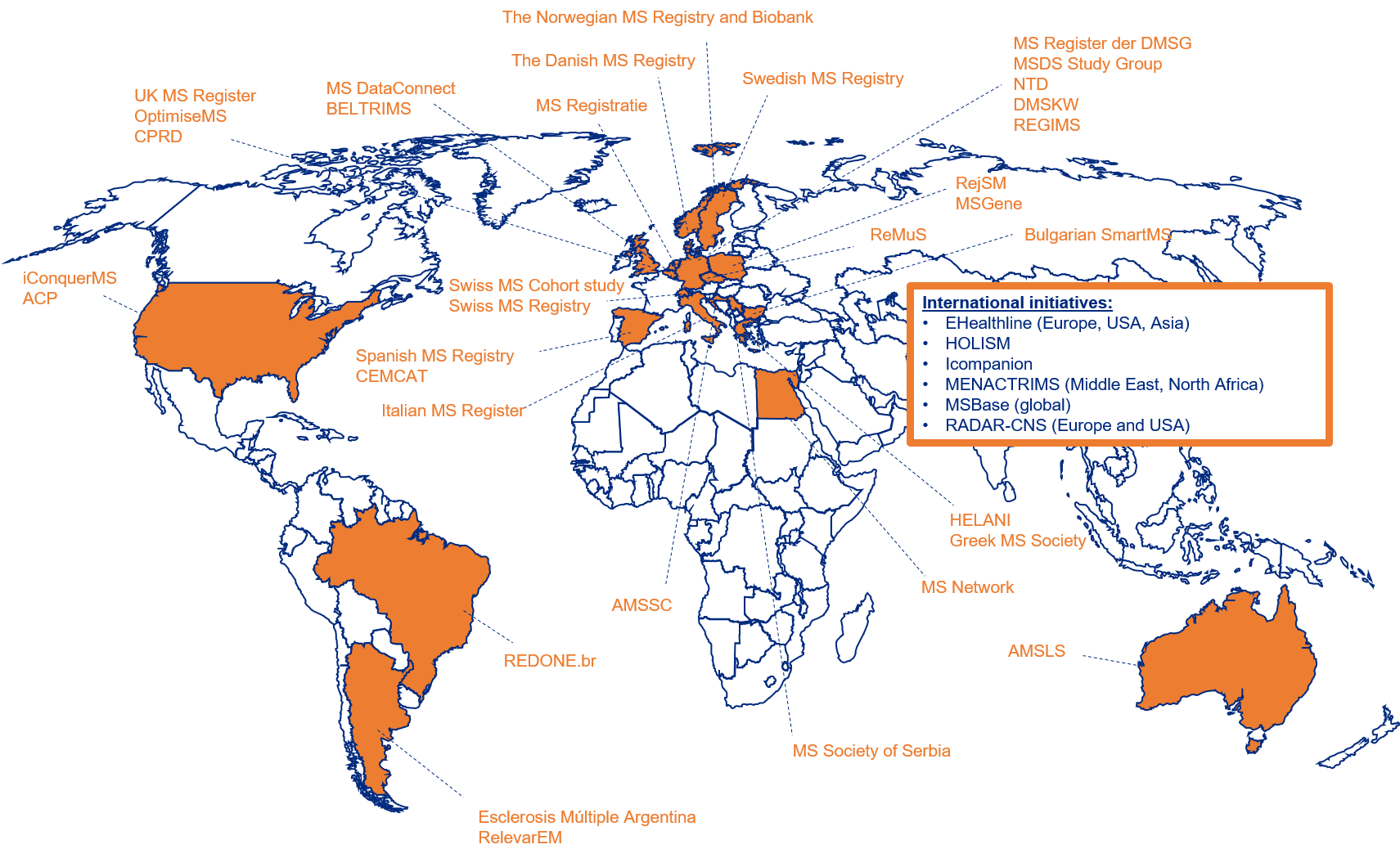
There are many existing and arising real-world data (RWD) sources in MS. They differ in purpose, maturity and collect different variables. The MSDA launched the MSDA Catalogue in 2019. It allows end-users with particular study requirements or research questions to browse metadata profiles of MS real-world data cohorts. Detailed cataloguing of MS RWD is a first and useful step towards reducing the time needed to discover real-world MS datasets and therefore to facilitate collaborative research.
The MSDA Catalogue is available worldwide (link) and collects descriptive information on governance, purpose, inclusion criteria, procedures for data quality control, how and which data is collected. The current cataloguing procedure is performed in several manual steps and only consists of metadata / descriptive data and therefore has no privacy concerns. The MSDA Catalogue used to be hosted by the European Medical Information Framework (EMIF), which was organised around disease communities (MSDA was one of the communities in the EMIF platform). Recently, the MSDA Catalogue got its own personal platform, under the umbrella of the EMIF platform, and today, there are more than 40 globally spread registries / initiatives that share descriptive data with the MSDA Catalogue:
The MSDA platform (Catalogue), can be accessed via https://msda.emif-catalogue.eu/login. However, to view the MSDA Catalogue you will need to create an account first (this only has to be done once, and people who already joined the MSDA community via EMIF don’t need to create a new account).
If you are interested in becoming part of the MSDA Catalogue, or you would like to receive additional information, or if you have questions or are facing issues with the Catalogue, do not hesitate to contact Lotte Geys (lotte.geys@uhasselt.be).